Don’t giggle. OK, go ahead and giggle a little. Get it out. Then keep reading. As I researched this topic — medical marijuana for MS — I found that about half of my friends responded with a half-baked smile, while the other half spouted statistics about billions in tax revenues.
Call me a party pooper, but I actually don’t care about getting high or tax revenue, though I hugely appreciate any issue that acts as a lever to legalize marijuana.
I care only about staying mobile and healthy with MS for as long as possible. Forever would be nice. Since I live in the Commonwealth of Virginia, where marijuana is fully illegal as of 2018, I also really care that I could lose my job (and health insurance) if a random drug test finds an illegal substance in my blood, even with a prescription. Even in states where marijuana is legal — for example, California, Colorado, Michigan — legal complications are ugly and serious.
If you read my other blog posts, you know that my wellness strategy for MS is based on a whole-foods, plant-based diet, but I am constantly searching for additional science-based approaches to keep me healthy for as long as possible.
The topic is so complex that I am incapable of explaining most of it succinctly or accurately in my humble little blog. The agenda of the Second National Cannabis Policy Summit in April 2018 brought together representatives of Congress, The Brookings Institution, the Attorney General of DC, doctors, lawyers, civil rights activists, journalists, lobbyists, and others to discuss the most pressing policy challenges and opportunities. For the mildly curious, this 11-card deck provides a nice overview, and for the more curious, this Cannabis 101 series of short videos answers a wide spectrum of questions. Consumer Reports provided a simple set of guidelines on how to shop for CBD.
One aspect that directly impedes my ability to understand how the heck medical marijuana could benefit (or harm) my health with MS is Schedule 1 classification. According to the Controlled Substances Act signed by Richard Nixon in 1970, the federal penalty for trafficking less than 50 kilograms of marijuana, a Schedule 1 drug, is no more than five years in federal prison and up to a $250,000 fine for the first offense. Ouch. Schedule 1 drugs (e.g., heroine, marijuana) are regarded to have no medical value.
One doctor, Mikhail Kogan, who happens to be my primary care physician, is a leader in the campaign to increase research into the efficacy of medical marijuana. As the medical director of The George Washington Center for Integrative Medicine, Dr. Kogan is optimistic but cautious about the powerful compounds in marijuana.
So what about MS? I was surprised at the unequivocal terminology used by a few very reputable sources. Dr. Kogan pointed me to a 2017 report by the National Academy of Sciences, Engineering, and Medicine, described as one of the most comprehensive studies of research published since 1999 on the health effects of recreational and therapeutic cannabis use in a spectrum of illnesses, from cancer to MS to HIV to depression, etc. You can download free chapters (including Chapter 4, Therapeutic Effects, referencing MS) or buy the $65 full report. Conclusions included:
Conclusive or substantial evidence that oral cannabis or cannabinoids are effective for improving patient-reported multiple sclerosis spasticity symptoms (oral cannabinoids); and
Moderate evidence of improving short-term sleep outcomes in individuals with sleep disturbance associated with…[lots of other things and] multiple sclerosis (cannabinoids, primarily nabiximols).
The report highlights the need for a national cannabis research agenda that includes clinical and observational research, health policy and health economics research, and public health and public safety research.
There is also the 2017 article, Cannabis and MS – The Way Forward, published in Frontiers of Neurology of the U.S. National Institutes of Health. The article can be summed up with the following excerpt:
…There are many open questions regarding cannabis use [as an MS therapy], including optimal strains, frequency of use, other dosage questions, risks of long-term use, and which symptoms it effectively treats. These are all important questions in which the NIH and MS foundations should be interested. Unfortunately, up to date, NIH has not funded research grants on the benefits of cannabis in MS (10). Furthermore, there are no current research projects on cannabis funded by the National MS Society (11). Why is this the case? We hypothesize that while grant reviewers make the argument that randomized controlled trials (RCTs) are needed for this research, few if any are possible in the current legal framework…
Nice. Bight me, legal framework. And while I am at it, the U.S. National MS Society can bight me too, since they should be out in front, advocating for sensible legal reform benefiting people with MS. The article also explains the twisted institutional knots and bias that obstruct serious scientific research to answer a long list of questions.
In the margin of this article, several links are provided to other articles on MS and medical marijuana. In the same publication, this 2018 article — Cannabidiol (CBD) to Improve Mobility in People with MS — caught my attention. A quick summary:
…Based on the following considerations, it is our opinion that CBD supplementation maybe advisable for people with MS (PwMS) to reduce fatigue, pain, spasticity, and ultimately improve mobility…It is clear that more research is needed. However, because of the safety of CBD and if the concerns listed above are accounted, we are in the opinion that we already have some good reasons to believe that CBD enriched cannabis is useful to improve the mobility of PwMS.
This article also looks at key considerations, including risk of addiction, contraindications, labeling inaccuracies, and the possibility of testing positive for marijuana.
I am left with a whiff of hope and a mix of disgust and sadness that potential solutions are stuck behind the bureaucratic stupidity of Schedule 1 classification. Regardless of these barriers, the 2017 article mentions that approximately 50% of people with MS already use cannabis, and the 2018 article mentions that 66% of people with MS use cannabis. Time matters for people with MS. Time waiting for reform is time lost to disability. Screw that. No seriously, screw it.
Dr. Kogan prescribed medical marijuana for me in early 2018 in the form of CBDa, one of many, many strains of marijuana, which I take in a tincture (droplet) form in a base of MCT oil to aid absorption. This mix is super tasty. Less tasty, flaxseed oil has the highest Omega-3 content, which is recommended – unrelated in any way to marijuana – by Overcoming MS, but flaxseed oil requires constant refrigeration.
A few questions that I asked when considering whether and how to take CBDa follow:
Dose? Thanks to the scarcity of large-scale studies caused by institutional obstacles, it’s currently impossible to say. In this podcast, Dr. Kogan speaks with Dr. Donald Abrams, one of the world’s leading clinical researchers on medical cannabis and an integrative oncologist at the University of California San Francisco, about the total lack of nomenclature for dosing. In DC, where medical marijuana is legal, Dr. Kogan is not legally able to advise patients on what strain of cannabis to take.
As an impatient patient of MS, I ask: how fricken stupid is that?
Kogan asks, “How do we guide our patients?” Dr. Abrams responds, “That’s the $64,000 question. Go to the dispensaries, tell them what medical condition you have; they deal with other patients who have what you have. See what they recommend.”
Generally, the guidance is to start low and go slow. After more than six months, I take roughly two full droppers (5 mg x 2) of CBDa in the morning and evening.
Source? I get my CBDa from a person recommended to me by Dr. Kogan. He has been to this person’s production facilities in Vermont, where the hemp plant is grown organically. The producers than send every batch to MCR Labs in Massachusetts.
Safety? MCR Labs produces a report on the contents of the tinctures that my source produces and sends to me. I provide a screen shot below of the batch sent to me in February 2018. Notice that THC, the psychoactive portion, is at 1.75%. In other words, an illegal substance could show up in my blood test, but the amount would be so low that I could pass the test. However, for whatever crazy reason, it could generate questions/concerns, and so my source told me bluntly, “it is a risk” that my employment could be terminated.
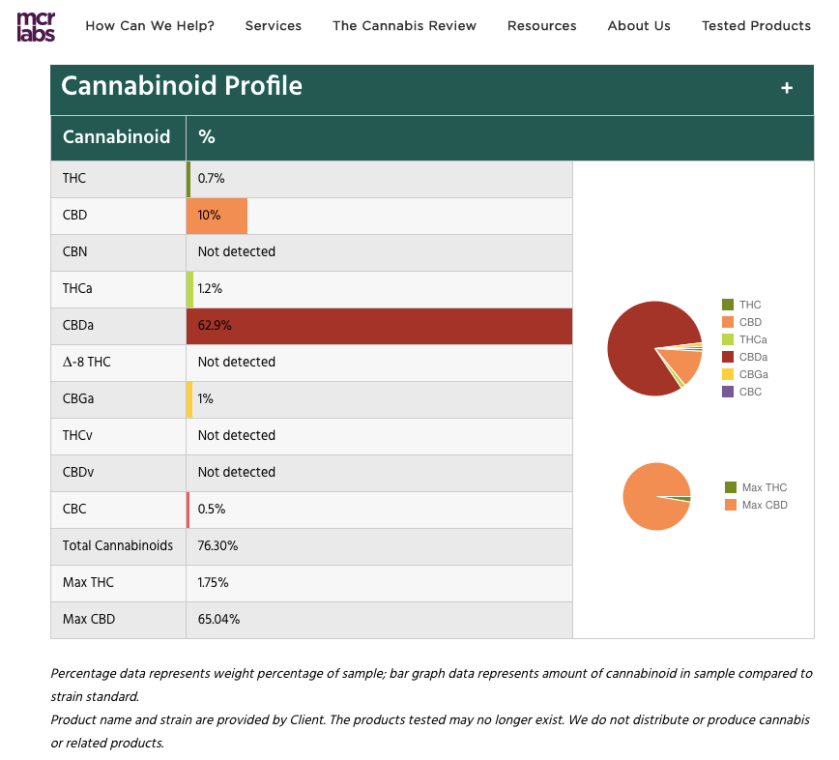 Price? I pay (in 2018) $50 per 300 mg bottle, which lasts roughly a month (or two; my adherence has been imperfect, even with no official guidance on dosing). No, it is not covered by insurance.
Price? I pay (in 2018) $50 per 300 mg bottle, which lasts roughly a month (or two; my adherence has been imperfect, even with no official guidance on dosing). No, it is not covered by insurance.
Why in tincture form? The veins under our tongues bypass the digestive system, specifically the liver and metabolites, which could mitigate the impact of powerful compounds, and make a bee-line for our brains. Since MS is a disease of the central nervous system, a tincture gets the CBDa to its destination more directly.
How does CBDa make me feel? You know that awesome feeling after you blink? No? Right. That’s how euphoric I feel after taking CBDa. I get no high — zero, zip, nada. That’s what most people seem to want to know. As I said, I do love the flavor. It’s like you have the freshest pine tree under your tongue for a couple minutes. Wow?
So why do I bother with CBDa, given that it ain’t cheap? Several reasons:
First, it is neuro-protective and possibly neuro-generative, so I consider CBDa an additional insurance policy — on top of a meticulously healthy diet, exercise, meditation, and other critical daily routines — for the care and maintenance of my central nervous system, slightly bedraggled after nearly a decade of doing nothing beyond MS drugs after my MS diagnosis in 2003.
Second, medical marijuana has demonstrated clinical benefits and promise in U.S. government-funded studies for MS (linked above) and cancer*, and the FDA approved medical marijuana for pediatric epilepsy**. How can the FDA recognize the therapeutic value, while the definition of a Schedule 1 drug is that there is no medical value? The U.S. government needs to get its ducks in a row.
I am not willing to wait for the political knot to be untangled; I am willing to risk my employment because I can get another job, but I can’t get another nervous system. I feel sorry for anyone whose job is to deny someone access to potential therapies for pain, seizures, immobility, spasticity, etc.
Third, I share with a bit of embarrassment, but in the spirit of truth made easier with an anonymous blog: CBDa seems to have saved me from wicked night sweats, which could result from a spectrum of causes (e.g. peri-menopause, cancer, etc), but which are definitely a symptom of MS rarely discussed in polite company. I had them almost nightly for about a year; now with CBDa, they are an occasional whisper of their former raging strength. That’s the only (but very welcome) physical change I have noticed since starting CBDa, but it makes me wonder what other benefits I could be accruing.
Plant versus pharmaceutical? Dr. Kogan said that there have been so few large-scale studies on medical marijuana that any commercial supplier that says they know the right dose/strain for specific ailments is not being honest.
For me, as long as I have a safe source of medical marijuana, I prefer the basic plant. Beyond my fascination with the science behind the power of a whole-foods, plant-based diet, I learned something mind-blowing: Have you ever heard of your body’s EndoCannabinoid System (ECS)? I had not. Thanks to Wikipedia, I learned that the ECS is a biological system composed of endocannabinoids, which are endogenous lipid-based retrograde neurotransmitters (huh?) that bind to cannabinoid receptors and receptor proteins that are expressed throughout the mammalian central nervous system, including the brain and peripheral nervous system.
What actually is a cannabinoid [kuh–nab–uh-noid]? Simply, it is any of the chemical compounds that are the active principles of marijuana, the dried leaves and and female flowers of the hemp plant (Latin: Cannabis sativa). Cannabinoid = hemp = marijuana = cannabis. It’s far more complicated than that, but my non-scientific brain needs the Dummies version.
So, our bodies have receptors for the cannabinoid compounds in the hemp plant. That factoid brings renewed significance to the power of plants. Wow.
In conclusion, I am not endorsing medical marijuana for you, and I certainly do not feel it is a stand-alone silver bullet. This blog is an attempt to explain why I include it in my large toolbox for wellness. Perhaps someday, reputable research institutions can conduct rigorous, large-scale, double- and triple-blind studies on the bio-mechanics of cannabinoids for MS. Then, if the evidence continues to show efficacy, I hope that insurance companies will make medical marijuana available and affordable to everyone with MS. I can’t imagine how many years that will take, but I am not waiting. Until then, I’ll just enjoy my tasty pine-tree flavored treat.
~~~~~
*Excerpt from link: To summarize, cannabis and cannabinoids are useful in managing symptoms related to cancer and its treatment. Exciting preclinical evidence suggests that cannabinoids are not only effective in the treatment but also in the prevention of chemotherapy-induced peripheral neuropathy. Cannabinoids could be synergistic with opioids in the relief of pain. The safety profile of cannabis is acceptable, with side effects that are generally tolerable and short-lived. Preclinical data suggest that cannabinoids could have direct anti-tumor activity, possibly most impressive in central nervous system malignancies. Clinical data about the effects of cannabis concentrates on cancer are as yet unavailable. Oncologists could find cannabis and cannabinoids to be effective tools in their care of patients living with and beyond cancer.
**Excerpt from link, “…The FDA needed to see solid evidence, meeting rigorous criteria, before approving any cannabis-derived drug for medical therapy. And [this] is the first drug to have achieved these high standards.”

There are a number of studies that show other negative health consequences short and long term of marijuana use. So, you are right that more research is needed. Can control trials not be carried out in states where the substance is legal? It seems that marijuana, as it is not the silver bullet to cure or relieve MS, could be dangerous for you, or at least could have negative effects on the mind and body that you are working so hard to protect. Have you exhausted all other avenues, finding that the relief you get from marijuana cannot be attained from other treatments? What is it about the marijuana protocol that makes you so fiercely advocate it for MS patients? I am interested to know more. Will keep linking to more of the research to which you guide us. Thank you for your courage in sharing your personal story.
LikeLike
I edited-out a section where I noted that it is extremely difficult to get a license to study a Schedule 1 drug, partially bc, by definition, Schedule 1 drugs have no medical value, which of course the FDA challenged with its approval for use of cannabis for pediatric epilepsy. The 2017 Frontiers in Neurology article that I linked in this blog explains: “To investigate the effects of cannabis, a researcher must win approval from the Food and Drug Administration (FDA), Public Health Service, Drug Enforcement Administration (DEA), NIDA, and the Institutional Review Board (IRB). For comparison, to research heroin a researcher must get approval from FDA, DEA, and IRB. Investigators are required to possess a DEA schedule I license in order to perform interventional studies, which is the biggest challenge. Currently, only few laboratories in the United States have a schedule I license. Therefore, it is unrealistic to require cannabis research to be performed in the same manner as traditional medications as long as cannabis remains Schedule I.”
LikeLike